About us

The Trials@Home consortium explores the opportunities of moving clinical trials from the traditional clinic setting to the participant’s immediate surroundings. These so-called Decentralised Clinical Trials (DCTs) make use of new, digital innovations and enable participants to visit a clinical trial centre less frequently, if at all.
This makes it easier for larger, more diverse and remote populations to participate in clinical trials. These trials are expected to be conducted faster, more efficiently, and provide results that are more representative, because the data is collected in the daily context of the participant (Real World Evidence). The research to be conducted includes an inventory and evaluation of existing and new techniques for use in DCTs as well as a pan-European pilot trial.
Co-creative, multi-stakeholder approach
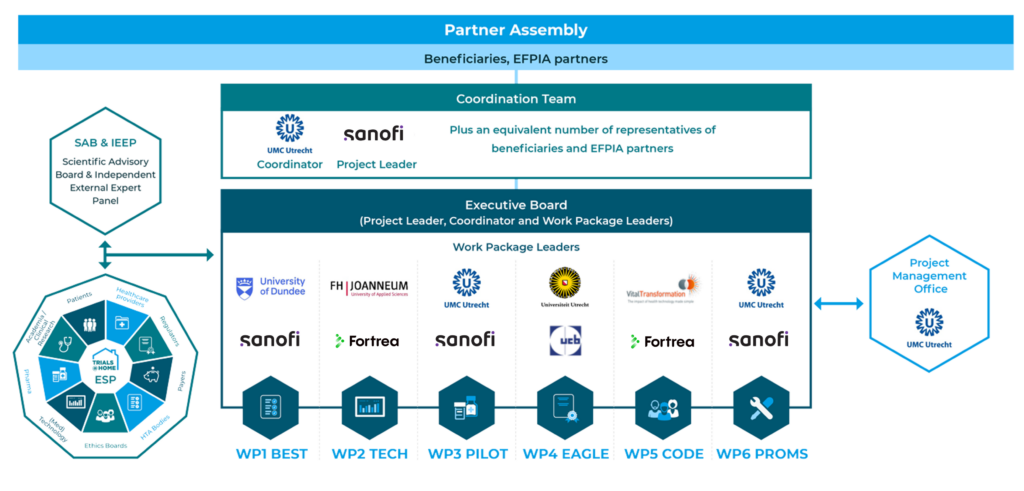
Trials@Home is an IMI (Innovative Medicines Initiative) funded research project that follows a co-creative multi-stakeholder approach where academic partners, Small and Medium-sized Enterprises (SMEs), private foundations, and EFPIA partners will work together with other stakeholders from across the medical, technological, regulatory, ethical and social aspects of DCTs, with a common goal to develop concrete and practical recommendations, and pilot tools supporting widespread acceptance and use of DCTs in Europe.
The University Medical Center Utrecht and Sanofi are coordinating this €40 million research project which will run for a total of six years and three months (five years and 15 months extension).
Our partners and the assembly