RADIAL Country selection process
To select the countries where the pan-EU RADIAL pilot study will be conducted, a multi-step approach was used while considering a good spread across Europe and health care systems, the legal and regulatory framework in those countries and the operational aspects such as potential trial participant recruitment and study start-up timelines.
The steps included:
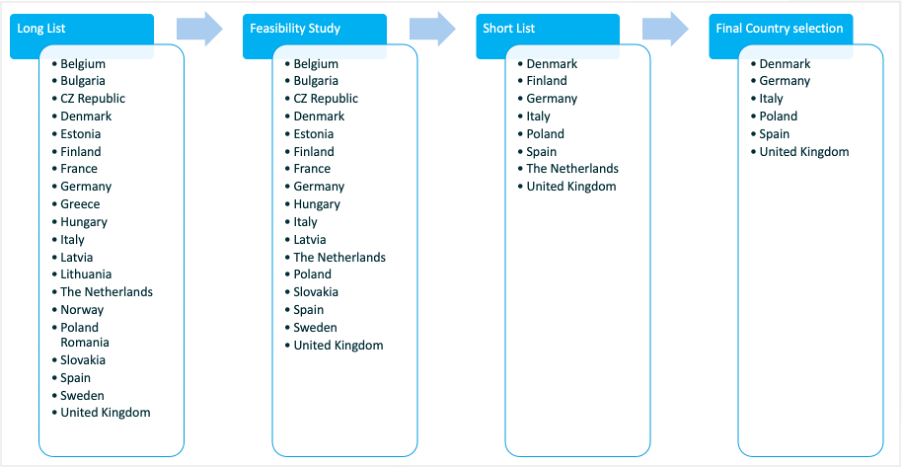
- The creation of an initial long list of 21 potential countries by Work Package 4 (WP) EAGLE (see Deliverable 4.1) of which ten were selected based on either prior positive experience or expectations for recruitment of the target study population.
- As a result of the adaptation of the initial list based on potential knock-out criteria (i.e., prevalence of Type 2 Diabetes; start-up timelines; acceptability of some decentralised trial elements such as eConsent or home delivery of IMP; digital literacy based on gathered intelligence from consortium members and EAGLE review; and IMP reimbursement), four countries were dropped.
- A feasibility study was then conducted on site-level in the remaining countries to identify the Selected countries list.
The site-level feasibility process included a questionnaire to known diabetes clinical trial sites in countries which were part of the ‘Feasibility Study’ list.
Based on the outcome of the feasibility study, the following country allocation was proposed to, and accepted by the steering committee: Denmark, Finland, Germany, Italy, the Netherlands Poland, Spain, and the UK.
After further deliberation by the steering committee to take into account the available budget, the Netherlands and Finland were dropped from the country list. As a result, the RADIAL pan-EU study will run in 5 EU countries and in the UK.



This project has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement No 831458. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation programme and EFPIA.
Notes for editors – not for publication
If you would like more information, please contact the spokesperson at the UMC Utrecht.
Joris Prinssen: +31 6 2571 0234
press@umcutrecht.nl
